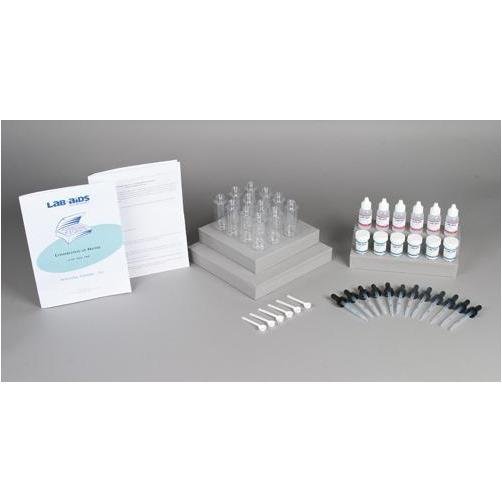
Lab-Aids: Conservation of Matter Kit
In this activity students investigate the difference between closed and open systems and confirm, the Law of Conservation of Matter. They conduct and compare a chemical reaction taking place in a closed system with one occurring in an open system. They observe that while the mass of a gas-producing reaction decreases in an open system, it remains the same in a closed system. Their data is also used to reinforce the molecular nature of all matter, including gases.
Scientific Concepts:
- Matter is neither created nor destroyed during chemical reactions.
- Substances react chemically in characteristic ways with other substances to form new substances that have different chemical properties.
- In reactions, the starting materials-reactants-are chemically altered to form products.
Content List:
- 1 Teacher's Guide with MSDS
- 28 Student Worksheets and Guides
- 12 Reaction bottles with screw-on dropper top
- 6 30 mL bottles of 4M HCl (half filled)
- 6 15 g vials of NaHCO3
- 6 White scoops
Classroom Planning:
- To complete this kit requires one to two ~55-minute class periods
- Number of students: 24
You may also like
More from All STEMfinity Brands
Recently viewed